Pharmaceutical & Industrial Integration
Integrated Bio-Defense. Molecular-Level Microbial Neutralization for Advanced Materials.
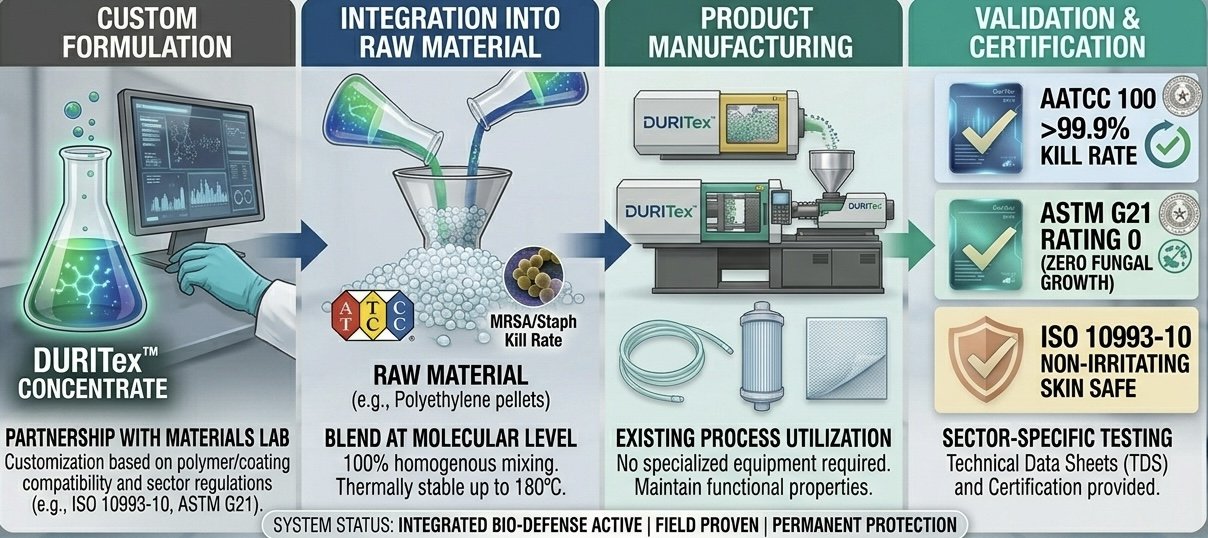
DURITex™ moves beyond coatings and topicals to offer customizable, high-concentration antimicrobial technology designed for molecular-level integration into the manufacturing value chain. We partner with product engineers and materials scientists to infuse persistent antimicrobial properties directly into polymer systems, coatings, and specialized fluid management components, creating inherently self-sanitizing products.
The Molecular Integration Process
Mechanical Kill: A Permanent Transformation.
DURITex™ doesn’t leach; it forms a permanent molecular bond with the host material, transforming its physical surface at the atomic level.
Molecular Spike Formation: A long-chain organofunctional silane is integrated into the material matrix, creating microscopic, positively charged "spikes."
Electrostatic Attraction: Pathogens (bacteria, fungi, and viruses) are drawn to the spiked surface by opposing electrical charges.
Mechanical Rupture (Lysis): The spikes physically puncture the outer membrane of the microbe on contact, causing instant destruction.
Result: The host material itself becomes antimicrobial, preventing pathogen colonization and the development of antimicrobial resistance (AMR), for the life of the product.
Customized Sector Integration
Targeted Solutions for Critical Infrastructure.
DURITex™ is not a one-size-fits-all solution; it is a platform technology customized for specific industrial performance and regulatory requirements.
A. Pharmaceutical Manufacturing & Cleanrooms
Cleanroom Surfaces: Infuse epoxy floorings, wall panels, and HVAC seals to maintain sterile integrity and reduce bio-burden.
Fluid Handling: Gaskets, seals, and specialized tubing treated to prevent biofilm accumulation in high-purity water (WFI) systems.
Packaging Integrity: Integrate antimicrobial protection into primary or secondary packaging to ensure long-term sterile delivery.
B. Water Filtration & Treatment
Membrane Integration: Treat reverse osmosis (RO) membranes and filter media at the point of manufacture to inhibit biofouling.
Flow Efficiency: Prevention of microbial growth maintains optimal flow rates and extends filter lifespan.
C. Specialized Medical Devices
Implantables & Tools: Molecularly bond to medical-grade silicone, thermoplastics, and coatings to reduce device-related infections.
Sterile Touchpoints: Treatment of high-contact surfaces in surgical suites and patient monitors.
Validated Integration Performance
Key Integration Benefits
Broad-Spectrum Efficacy: Effective against bacteria, viruses, and diverse fungal strains (validated).
AMR Mitigation: Microbes cannot mutate to build resistance to mechanical destruction.
Non-Leaching: Guaranteed zero migration into pharmaceutical products or the environment.
Persistent Activity: Remains active for the functional life of the host material.
Thermal Stability: Stable in manufacturing processes up to 180°C.
Pilot Your Solution
Consult with an Integration Specialist.
DURITex™ technology is customizable to meet the specific compatibility and regulatory benchmarks of your polymer, coating, or specialized material. Partner with our materials science lab to develop and validate a custom formulation.